Chemotherapy: A Key Player in the Battle Against Pancreatic Cancer
No matter what stage of pancreatic cancer you have, chances are good that chemotherapy will play a starring role in your treatment regimen.
“About 55,000 people are diagnosed with pancreatic cancer each year and only 25% of those are surgical candidates,” says Vincent Chung, M.D., medical oncologist at City of Hope in Duarte, California. Which chemotherapy you and your doctor choose depends largely on the goals of treatment.
Chemotherapy Explained
Chemotherapy protocols are designed to stop cancer cells in their tracks, preventing them from growing, dividing, and spreading to other organs. The medications travel through the bloodstream where they can kill cancer cells throughout the body.
“More than half of all patients present with metastatic disease, so the goal of treatment is to control the cancer so patients have less pain, more energy, and ultimately live longer lives,” says E. Gabriela Chiorean, M.D., Professor of Medicine and Clinical Research Director of Gastrointestinal Medical Oncology at University of Washington and Clinical Director of Gastrointestinal Medical Oncology at Fred Hutch Cancer Center.
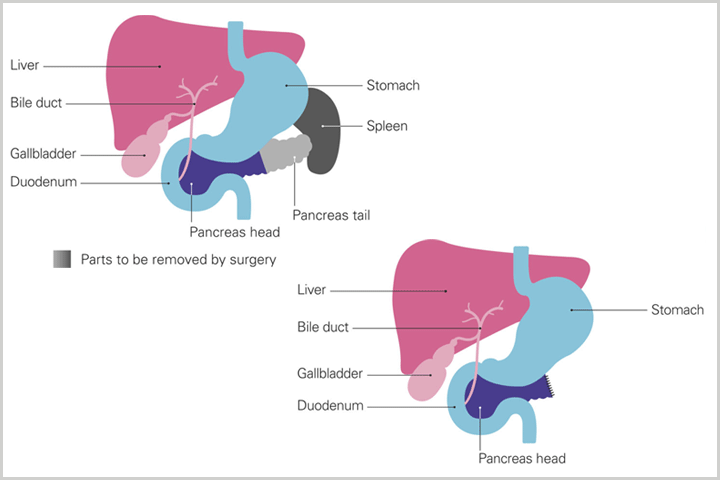
Some patients receive neoadjuvant chemotherapy—chemotherapy before planned surgical removal of the tumor—to shrink the tumor. There are other patients who are on the cusp of being a surgical candidate, with tumors that could be removed if made smaller. For those patients, neoadjuvant chemotherapy can be given before being re-evaluated for surgery, to see if the tumor is small enough to make surgery possible. In both cases, neoadjuvant chemotherapy can increase the odds of successful surgery, especially where the cancer is close to veins and arteries. Surgical patients very often receive chemotherapy after surgery, called adjuvant chemotherapy, to decrease the chances of a recurrence of the cancer.
Chemotherapy Regimens for Pancreatic Cancer
The Food and Drug Administration (FDA) has approved four chemotherapy drugs for the treatment of pancreatic cancer: gemcitabine (brand name Gemzar), nab-paclitaxel (brand name Abraxane), liposomal irinotecan (brand name Onivyde), and 5-FU (fluorouracil). NCCN (National Comprehensive Cancer Network) guidelines also include treatment with oxaliplatin in combination with 5-FU based chemotherapy, and the drug combination FOLFIRINOX. Each of these treatments have varying levels of efficacy and toxicity.
“If someone has early stage I or stage II disease, where the goal of treatment is potentially curative, we tend to be more aggressive with our chemotherapy choices,” Chung says. Here’s how it breaks down:
5-FU: Before 1995, fluorouracil (5-FU) was the standard treatment for advanced pancreatic cancer. In trials using CT assessment of tumor response, the reported response rates for single-agent 5-FU ranged from 0% to 19%. Now 5-FU is typically used in combination with other therapies.
Gemcitabine: In 1996, the FDA approved gemcitabine for the treatment of pancreatic cancer that doctors can’t remove surgically. A 2007 report showed benefit in preventing recurrence after surgery.
Nab-Paclitaxel: In September 2013, the FDA approved nab-paclitaxel for use, in combination with gemcitabine, as a first-line treatment for metastatic pancreatic adenocarcinoma.
Liposomal Irinotecan: In October 2015, the FDA approved liposomal irinotecan in combination with 5-FU for metastatic pancreatic adenocarcinoma that progressed after treatment with gemcitabine.
Platinum-Based Agents: According to a February 2019 study published in the Journal of Clinical Oncology, platinum-based agents, such as cisplatin and oxaliplatin, can greatly improve survival among patients with BRCA1 or BRCA2 mutations. Since these mutations impact the ability of DNA in cancer cells to repair itself, they are more susceptible to platinum chemotherapy.
FOLFIRINOX: Studies from 2011 showed the combination of 5-FU with irinotecan, leucovorin, and oxaliplatin (FOLFIRINOX) had the largest benefit for patients with metastatic pancreatic cancer. “This is the gold standard treatment for patients who are fit, and able to tolerate this regimen,” Chiorean explains.
“FOLFIRINOX is the most difficult and complicated regimen—in part because you have to go home with a pump—but it’s also the most effective,” notes Chiorean. “Even in locally advanced cancer that cannot be removed upfront, data show FOLFIRINOX leads to a chance for resection rates between 30 and 60 percent.”
In 2018, FOLFIRINOX also showed the best efficacy to prevent cancer recurrence after surgery and is therefore widely used post-op. In a 2018 article in the New England Journal of Medicine, researchers reported an increase in median overall survival after surgery with FOLFIRINOX when compared with gemcitabine.
The challenge: FOLFIRINOX is quite toxic. Patients often experience digestive issues, nausea, vomiting, diarrhea, and cumulative and sometimes irreversible neuropathy. For these reasons, doctors typically reserve FOLFIRINOX for patients who are healthy enough to tolerate the side effects. Even so, people typically get chemotherapy in sequence. Chiorean explains, “So, if you start on one regimen with 5-FU based chemotherapy, you’ll eventually switch to the other with gemcitabine and nab-paclitaxel.”
Chemotherapy on Trial
For patients with any stage of pancreatic cancer, clinical trials may be the best avenue. Not only do trials offer early access to the most leading-edge treatments, they also ensure you’re closely monitored throughout your treatment regimen. The end result: improved treatment options and better outcomes.
One of the most impactful research discoveries in the past year, however, has been the benefit observed from PARP inhibitors for metastatic pancreatic cancer patients with BRCA1 or BRCA2 gene mutations who initially responded to platinum chemotherapy. “Genomic profiling is now routinely done in tumors which are advanced both locally and metastatic and finding gene alterations may open the door to molecularly-targeted therapies or immunotherapies, and therefore more treatment options,” says Chiorean.
Managing the Side Effects of Chemotherapy
Chemotherapy attacks all dividing cells, whether they’re healthy or cancerous—and that means it can come with significant side effects. Healthy cells that rapidly divide, such as skin, hair, bone marrow and blood cells, and the cells in the reproductive and digestive tracts, are at greatest risk of damage.
Oncologists give chemotherapy in cycles, so patients have an opportunity to recover from the side effects of treatment, including:
- Changes in taste
- Fatigue
- Diarrhea and abdominal cramping
- Hair loss
- Hand-foot syndrome (tenderness, dryness, and peeling of the palms and soles)
- Low blood counts (white blood cell, red blood cell, or platelets)
- Loss of appetite
- Nausea and vomiting
- Mouth sores
- Nail changes
- Neuropathy (numbness and tingling in the hands and feet, and sometimes other areas of the body)
“It’s important to talk to your doctor about any side effects you’re experiencing,” says Chung, who encourages patients to keep a daily log.
Suffering from debilitating nausea from day 3 through day 5 of treatment? Your doctor can prescribe anti-nausea medication for day 2 during the next round of treatment. Experiencing severe cold sensitivity and neuropathy? Your doctor may decide to reduce the dose or discontinue oxaliplatin.
“We have choices with chemotherapy, so we can modify treatment depending on the side effects you’re experiencing,” says Chiorean. “Even the schedule can be adjusted to fit the patient’s lifestyle.” In every case, conversation and careful planning can help you sidestep troubling side effects and better tolerate treatment.