AI Platform Reveals Hidden Biology of Tumor Formation

If you’re of a certain age, your first encounter with artificial intelligence (AI) was probably a Saturday afternoon matinee watching Stanley Kubrick’s 1968 epic “2001: A Space Odyssey.”
In the movie, the breakout star was a computer named HAL 9000 that didn’t want to be unplugged. Fast forward to the early1980s and AI controlling the world of “Blade Runner,” while in the Terminator movies an advanced algorithm called Skynet wants to eliminate humanity.
In real life, however, AI is a lot more benign. After all, it has become part of our lives, whether we talk to virtual assistants, such as Siri or Alexa, or use GPS to navigate driving routes. These technologies all use some form of AI to make life easier.
But the power of AI also has tremendous applications in medicine. AI is currently being used to assist in clinical decisions and support as well as analysis of CT scans, X-rays, MRIs, and other images for lesions or other findings. In the near future, AI may also assist discovery scientists in answering some fundamental questions about the once hidden biology of tumors.
A New Platform Called CODA
AI is central to efforts at the Johns Hopkins Center for 3D Multiscale Cancer Imaging, a partnership with Yale University that was founded by Denis Wirtz, Ph.D., professor of chemical and biomolecular engineering, vice provost for research at Johns Hopkins, and a core researcher at Johns Hopkins’ Institute for NanoBioTechnology.
Laura Wood, M.D., Ph.D., Center co-founder, pathologist, and pancreatic cancer researcher, has joined forces with colleagues from disciplines as varied as oncology, bioengineering, mathematics, and computer science to develop an AI-assisted computational platform called CODA. The platform was created by Ashley Kiemen, Ph.D., a postdoctoral fellow at the Johns Hopking Whiting School of Engineering, and Pei-Hsun Wu, Ph.D., M.S., an associate research professor at Johns Hopkins’ Institute for NanoBioTechnology.
The group will try to figure out the answer to a question that has been plaguing scientists: How exactly do tumors form? They hope the answers they find may lead to earlier detection of pancreatic cancer, which, despite incrementally increasing survival rates, remains one of the most lethal of all cancers.
“Pancreatic cancer is a terrible disease that needs significantly better treatments and earlier detection,” says Wood, associate professor of pathology and oncology at Hopkins Medicine, director of gastrointestinal and liver pathology, and associate researcher at the Institute for NanoBioTechnology. “So what we need to do is help answer questions about treatment resistance and find ways in which this disease can be found earlier.”
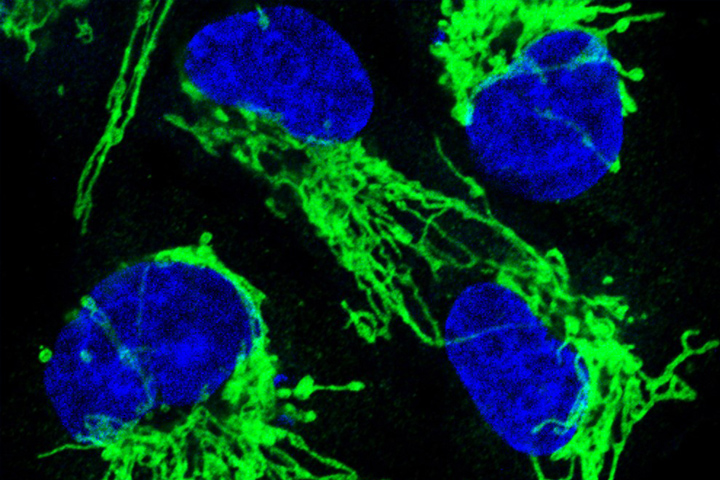
Wood explains that the CODA system allows researchers to produce 3D models by assembling sections of tissue samples so incredibly small “that we see the actual anatomy of each cell.” And because of AI’s ability to handle big data, “we can assess extremely large volumes of tumors,” she adds. “We believe this will give us a revolutionary ability to see, for example, the precise location where cancer cells enter the bloodstream to initiate metastasis.”
Pathology can be painstaking work. But CODA performs slide assembly quickly, and it also creates an algorithm that can quantify certain attributes of the tissue, like the number of cells it contains as well as other important data about the tissue that scientists “had no way of seeing before,” says Wood.
“We’re seeing microanatomy, the vasculature, and we see them in such a way that the new information can potentially aid in interception, diagnosis, treatment. We aren’t there yet, but I know we will be,” she adds.
Studying PanINs
In science-speak, tumor formation is called tumorigenesis. It’s a very complex and dynamic process by which a tumor forms, becomes cancerous, and eventually spreads to other parts of the body. Wood and colleagues are currently testing CODA’s mettle on a specific type of lesion called pancreatic intraepithelial neoplasia (PanIN), a type of microscopic lesion that can develop into pancreatic cancer in some cases. Pathologists have recognized precursor lesions in the pancreas for more than a hundred years. In fact, PanINs have been reported in 16 to 45 percent of pancreas tissues that don’t harbor a cancer.
According to Wood, PanINs hold a key to potentially intercepting pancreatic cancer early. The premise behind cancer interception is to figure out why some healthy cells turn cancerous so strategies can be developed to literally interrupt that process of change from benign to malignant, as well as to stop the disease before it invades the body, she explains.
In a yet-to-be-published preprint study, Wood and colleagues looked at PanINs using CODA in a cohort of 38 so-called large pieces of normal pancreas tissue from surgical resection specimens. These “pieces” are akin to a small stack of quarters and contain up to one billion cells.
With CODA, the researchers were able to generate quantifiable models of large pieces of human pancreas with single-cell resolution. They found a mean burden of 13 spatially separate PanINs per a microscopically tiny piece of sampled tissue. They then extrapolated that finding to the entire pancreas.
“We were a little shocked to find about 1,000 PanINs in an entire pancreas,” Wood says. “What that means is that most of us are walking around with PanINs. But considering that PanINs are common and pancreatic cancer is rare that tells us that something is going on with those PanINs that become malignant.”
To help find out what, the researchers then performed targeted and whole exome sequencing of the samples and found that almost all PanINs contained mutations in the oncogene KRAS. However, no gene other than KRASwas altered in more than 20 percent of the analyzed PanINs. Some PanINs also have multiple KRASmutations. The researchers also discovered that PanINs are multifocal, most often arising from independent clones, which means they acquired their own unique set of mutations.
“This is one of the deepest looks at PanINs that has ever been done,” Wood says. “It’s shocking that this PanIN initiation process occurs independently hundreds or even thousands of times in the same pancreas.” What these initial results do, says Wood, is to begin to create a framework for research into the biological underpinnings of PanINs, which can then help in stratifying risk of progression.
Wood and colleagues, with the support of the Lustgarten Foundation, will continue their work in a study called 3D Analysis of Human Pancreatic Tissue to Improve Early Detection of Pancreatic Cancer. Another CODA project, funded by Break Through Cancer, will study treatment resistance and is called Demystifying Pancreatic Cancer Therapy.
“Going from knowing very little about the biology of PanINs to learning so much more is the first step,” says Wood. “This is really exciting and it’s very collaborative. If we can better understand the biological events that turn a PanIN into cancer, we can then develop methods of early detection and better targeted therapies And that’s when we can improve people’s lives.”