The ABCs of Genetic Testing
Pancreatic cancer, like all cancers, is a fundamentally genetic disease.
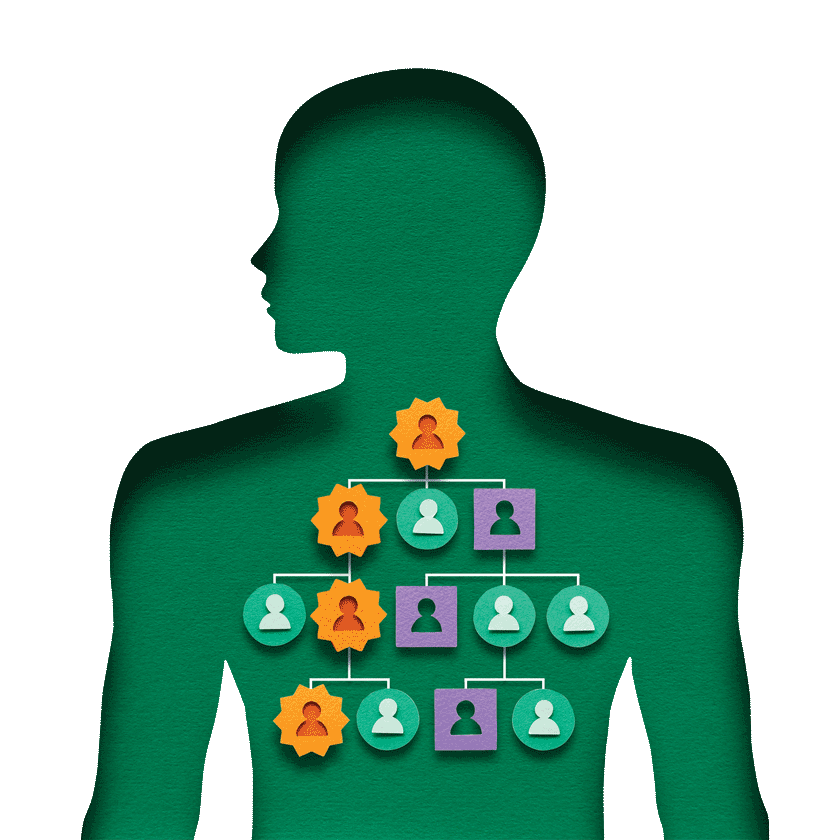
Most cases are caused by acquired mutations, but about 10 percent can be attributed to inherited mutations. These inherited mutations are the focus of genetic testing.
While the genetic mutations responsible for the majority of inherited, or familial, pancreatic cancers have yet to be identified, several pancreatic cancer genes have been established. Multiple gene panel tests can be used to investigate, among others, BRCA1, BRCA2, PALB2, CDKN2A, MLH1, MSH2, MSH6, PMS2, EPCAM, ATM, APC, STK11, PRSS1, and TP53.
But what does that alphabet soup of genes mean?
Tumor Suppressor Genes
Tumor suppressor genes normally keep cells under control by slowing down how often they divide (to make new cells), repairing DNA mistakes, or telling cells to die at the right time.
BRCA. BRCA mutations are the best-known, and may be familiar to the general public. Usually associated with breast and ovarian cancer, BRCA1/2 mutation carriers also have a mild to moderate level of risk for pancreatic ductal adenocarcinoma. This risk is slightly higher for those of Jewish heritage. While the risk for breast cancer is higher for those with BRCA1 mutations, it seems that an inherited defect causing a missing piece of DNA where BRCA2 should be found is the bigger risk in pancreatic cancer.
PALB2. PALB2 partners with BRCA2, as a protein that helps keep BRCA2 stable and facilitates DNA repair. Mutated PALB2 genes increase the relative risk of breast cancer and pancreatic cancer. The prevalence of PALB2 among families with pancreatic cancer clusters is estimated to be 4 percent.
CDKN2A. The CDKN2A gene, located on chromosome 9p21, encodes the p16 protein, an important cell cycle regulator. CDKN2A mutation is the main cause of familial melanoma, and an increased risk of pancreatic cancer in these families has also been noted. CDKN2A carriers may also face increased risks of sarcoma, lung, and breast cancer.
STK11. Mutations in the STK11 gene can cause the very rare Peutz-Jeghers syndrome, in which affected individuals develop polyps in their small intestines and pigmented spots on their lips and mouth. Individuals with Peutz-Jeghers syndrome have been shown to be at an increased risk of several cancers including esophageal, stomach, small intestine, colon, lung, breast, uterine, ovarian, and pancreatic cancer.
ATM. Inherited mutations in the ATM gene, known to cause the cerebellar degeneration syndrome ataxia telangiectasia, have also been found in 2-to-3 percent of people with familial pancreatic cancer.
Mismatch Repair Genes
Mutations in mismatch repair genes can also cause cancer. These genes are normally involved in repairing damaged DNA. When one of these genes isn’t working, cells can develop mistakes in their DNA, called microsatellite instability. This can lead to other gene mutations and eventually cancer.
MLH1, MSH2, MSH6, PMS2, and EPCAM. Lynch syndrome, the most common cause of hereditary colorectal cancer, can be caused by a series of mismatch repair genes: MLH1, MSH2, MSH6, PMS2 and EPCAM. Those with Lynch syndrome are also at higher risk for endometrial, ovarian, gastric, urothelial, skin, brain, and pancreatic cancers.
APC. Another colon-related syndrome that predisposes someone to pancreatic cancer is familial adenomatous polyposis (FAP). Caused by mutations in the APC gene, FAP syndrome is characterized by hundreds to thousands of polyps and tumors throughout the colon and other tissues, some of which are benign (adenomas) and others that become malignant.
PRSS1 and SPINK1. Those who suffer from chronic pancreatitis (inflammation of the pancreas) from an early age may also be at higher risk for pancreatic cancer. A rare inherited form of the condition, hereditary pancreatitis, is caused by mutations in either the PRSS1 (trypsinogen) gene or the SPINK1 (serine protease inhibitor) gene.
What Triggers Pancreatic Cancer?
A person who has a hereditary predisposition towards pancreatic cancer is considered to be at high risk of developing the disease. However, it is important to keep in mind that most pancreatic cancers—even in high-risk people—result from complex and progressive interactions between genes and environment. Things like diet, physical activity, alcohol and tobacco use, and any of a number of exposures, including toxins, pollutants, and sunlight, can influence gene activity. Scientists are also studying the effect of the inner body environment on cancer development and progression, such as the “tumor microenvironment” and gut bacteria in the “microbiome”.
Here’s how it happens. As we know, people have two copies of each gene in their bodies. A person born with an inherited mutation in one copy of a gene that predisposes to pancreatic cancer only has to acquire a mutation in the other copy of that gene for it to stop working. In addition, a triggering event, like a mutation in the KRAS gene, may also activate tumor development.
Researchers have found that these acquired mutations can accumulate through the combination of events like inflammation, exposure to toxins that damage DNA, and routine errors like failed DNA repair within cells.
Impact on Treatment Decisions
Knowing the state of your genes is not only important for risk assessment, it could also inform treatment decisions should you develop cancer. For example, tumors with genetic mutations in genes related to DNA double strand damage repair, such as BRCA1, BRCA2, PALB2 or ATM, have been associated with better responses to platinum-based chemotherapy and PARP inhibitors. Tumors with mismatch repair mutations may be the most likely to benefit from immunotherapy checkpoint inhibitors such as nivolumab or pembrolizumab.
Researchers are continuing to identify which genetic mutations respond better to certain treatments. This knowledge makes genetic testing of tumors important for both the risk of developing pancreatic cancer and treatment of that cancer if it occurs.
If you think you or family members may have one of the mutations mentioned in this article, read the story “Look Before You Leap: Genetic Testing for Pancreatic Cancer.”