In the News
When there's a news story featuring Let's Win, we want the world to know. Read the articles and watch the videos below to stay connected with the latest news about Let's Win.
37 Articles

Let’s Win in a Spectrum News NY1 Report for Pancreatic Cancer Awareness Month
Cindy Gavin and Dr. Allyson Ocean appeared in a Spectrum News NY1 report on a pancreatic cancer patient, for Pancreatic Cancer Awareness Month.

When Certain Chemotherapy Drugs Are in Short Supply
It’s been all over the news–some chemotherapy drugs are in short supply. Dr. Allyson Ocean explains a doctor’s options in these situations.

No Longer a Death Sentence
Dr. Allyson Ocean and Cindy Gavin discuss the mission of Let’s Win to raise awareness of the treatment options for pancreatic cancer.

PancChat Featured in a Journal Article
PancChat co-host Dr. Niraj Gusani has coauthored an article about the advantages of social media for oncology surgeons.
Should Cancer Patients Get the COVID-19 Vaccine? Yes!
Should pancreatic cancer patients get the COVID-19 vaccine? According to the experts, yes, and as soon as they can.

Families Working Together Against Pancreatic Cancer
Rob Greenstein discusses how Let’s Win helped his family after his sister Jackie was diagnosed with pancreatic cancer.
No Time to Wait: UK Pancreatic Cancer Patients Struggle with Treatment and Diagnostic Delays
Peter De Rosa of Pancreatic Cancer UK provides an update on how the COVID-19 pandemic has impacted pancreatic cancer patients in the UK.

Instagram Live with Dr. Allyson Ocean and Andrew Rakeman, Ph.D.
Dr. Allyson Ocean and Andrew Rakeman, Ph.D., got together on Instagram Live to discuss pancreatic cancer, risks, symptoms, and research.
COVID-19—An Update
What is the status of international and US travel? Which states have mask mandates? Are vaccines progressing? Read our COVID-19 update for more information.
Where We Are with COVID-19—A Wrap-Up
In a final COVID-19 Tip of the Week, pancreatic cancer patients need to continue social distancing, wearing masks, and washing hands frequently.

How COVID-19 Is Transmitted: The Latest Information
Virus in the air? Asymptomatic carriers? Surfaces? Pets? Find out the latest information about how COVID-19 is transmitted.
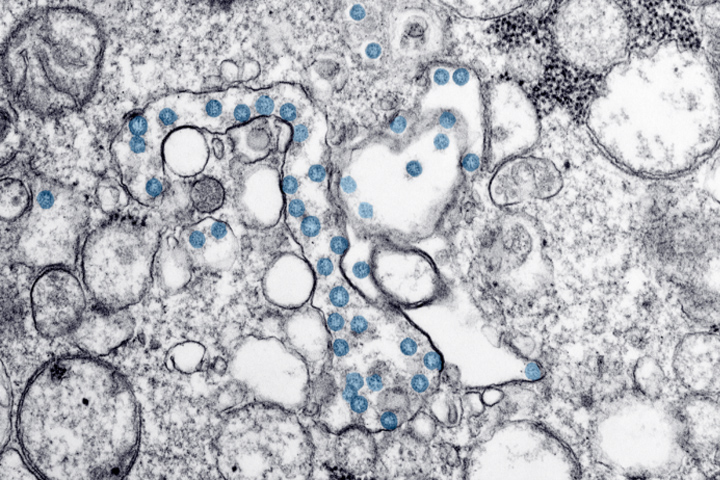
Confused about COVID-19 Testing? What You Need to Know
Confused by the information about COVID-19 testing? Our article explains the types of tests, whether testing is readily available, and how to get tested.

Keep Your Glasses from Fogging When Wearing a Mask
Here are some tips from doctors on how to prevent your glasses from fogging up when you wear your mandated mask for COVID-19.
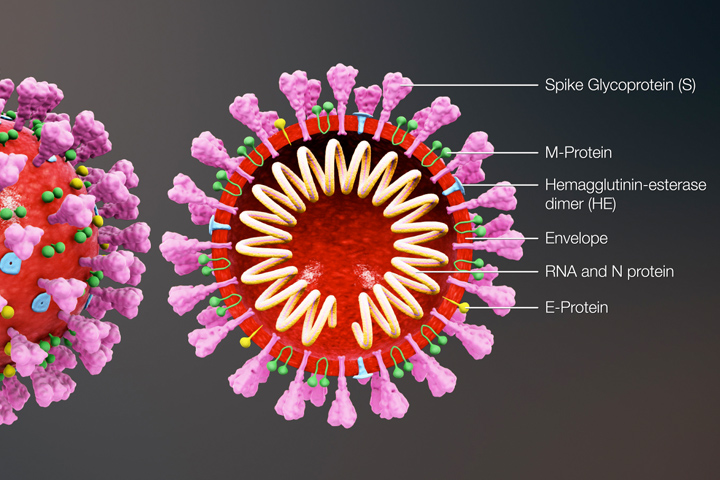
Reopening America: Guidelines For Pancreatic Cancer Patients
As states begin to reopen their economies, the threat of COVID-19 remains. Dr. Allyson Ocean has recommendations for pancreatic cancer patients.

Dos and Don’ts for Wearing Cloth Masks
Make sure you are wearing your cloth mask correctly to protect others from COVID-19 as you venture out more. And be sure to handle your mask properly after.