New Study Focuses On Supportive Oncology Care At Home

Let’s be honest. The symptoms of pancreatic cancer and the side effects of chemotherapy can be tough
For some newly diagnosed patients with borderline resectable tumors or locally advanced disease, neoadjuvant therapy is proving to be a game-changer. One such neoadjuvant approach uses FOLFIRINOX, a combination of the drugs leucovorin, fluorouracil, irinotecan, and oxaliplatin. Preoperative treatment with this regimen shows the potential to improve the odds of an R0 resection, meaning no cancer cells can be seen microscopically at the resection margin. Importantly, this correlates with improved median progression-free and overall survival.
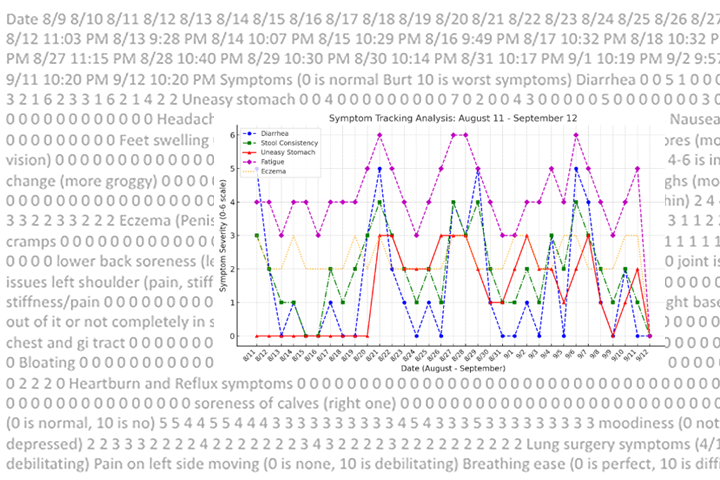
Yet the side effects of a FOLFIRINOX regimen can be debilitating, with many patients experiencing increased infection, nausea, vomiting, diarrhea, fatigue, fever, neuropathy, and loss of appetite. Taken alone, or together, these side effects often require urgent care or emergency department visits, and even hospital stays.
“Patients with pancreatic cancer who receive FOLFIRINOX often experience numerous side effects and a high symptom burden, and when these individuals need to be hospitalized or present to an emergency department, this adds an additional layer of burden. Thus, we are seeking to investigate novel interventions that address patients’ symptom burden, prevent hospitalizations, and ultimately improve the quality of life for patients with pancreatic cancer,” explains gastrointestinal oncologist Dr. Ryan Nipp of Massachusetts General Hospital (MGH), Boston.
Nipp is leading a new trial focused on providing supportive oncology care at home for patients with pancreatic cancer who are receiving neoadjuvant FOLFIRINOX. The goal of this pilot study is to determine the feasibility and acceptability of an intervention that targets a patient’s symptoms while delivering care to that patient in the home setting, with the ultimate goal of improving patients’ outcomes, including symptom management, an enhanced quality of life, and, hopefully, a reduction or elimination of the need for hospitalizations.
What Is Supportive Oncology Care at Home?
As part of this intervention, participants have access to remote daily patient monitoring with tablet computers. Each day, patients self-report their symptoms, such as diarrhea, constipation, nausea, fatigue, pain, and loss of appetite. Vital signs including blood pressure, heart rate, and temperature will also be monitored remotely with devices provided to patients. Weight is monitored weekly on a scale provided to the patient; the patient logs results via a tablet computer and is monitored by a hospital-at-home team.
Dr. Nipp and his co-investigators, working with Boston-based tech start-up Medically Home, have developed specific criteria geared to a cancer patient undergoing neoadjuvant treatment for pancreatic cancer. If a patient has new or worsening pain or concerning findings on their daily vital signs, for example, that would trigger the Medically Home team to contact the patient (via telephone), with the ability for clinicians to visit patients’ homes to address and manage any problems or other concerning issues. Importantly, the supportive oncology care with home intervention also entails regularly scheduled visits to a patient’s home to deliver intravenous fluids. Furthermore, the intervention also includes regular communication between the Medically Home team and oncology clinicians regarding the care delivered at home to ensure continuity of care.
“Although the idea of a ‘hospital-at-home’ care model is not new, this model of care has not been investigated within the field of oncology to date,” explains Nipp, whose research focuses on optimizing care delivery and outcomes for patients with cancer and their families. “Notably, patients with pancreatic cancer who are receiving neoadjuvant FOLFIRINOX represent a population with a particularly high likelihood of benefiting from this model of care. These patients are uniquely at risk for experiencing side effects of treatment and a high symptom burden from the cancer. They may become dehydrated quickly, and symptoms of fatigue, neuropathy, and lack of appetite can steadily worsen throughout the course of treatment. Consequently, the supportive oncology care at home care model represents a promising strategy to enhance patient outcomes, and we need to test the feasibility and acceptability of these types of interventions for patients with pancreatic cancer.”
A New Application for an Older Idea
Indeed, the idea of the so-called “hospital-at-home” isn’t new. It’s been in practice in certain European countries and Australia for years. Even in the U.S., health systems such as Mount Sinai in New York and Johns Hopkins in Baltimore have embraced the model. But it’s generally used for acute illnesses like pneumonia, exacerbations of congestive heart failure, and chronic obstructive pulmonary disease, among others. The research shows mixed results, but these programs tend to reduce readmission rates and costs and have positive patient satisfaction ratings. A 2019 article in the Journal of Clinical Oncology supports extending “hospital-at-home” programs to cancer patients.
“In our trial, patients will still receive their chemotherapy infusions in the clinic, that has not changed, and this represents a planned visit,” Nipp explains. “But the other type of visit we hope to prevent is the unplanned visit when, for example, a patient’s nausea or diarrhea reaches the point where the patient requires a hospital admission. We hope to avoid the need for urgent visits and hospital admissions by using a supportive oncology care at home intervention that utilizes state-of-the-art strategies to more closely monitor patients at their home and address issues earlier, before the problem escalates to the point necessitating a hospital admission.”
To that end, for example, Nipp and his team are taking a proactive approach with their supportive oncology care at home intervention: patients will receive fluids on a regular basis at home to avoid potential complications caused by dehydration, nausea, lack of appetite, and diarrhea. In addition, patients report their symptoms and vital signs daily, with pre-specified algorithms developed to address any concerning issues immediately, with the goal of preventing problems from escalating further.
Do Patients Like this Program?
Another goal of the study is to understand the acceptability of this intervention. Nipp and his co-investigators will explore the acceptability of this novel care model by asking patients, their caregivers, and also the clinicians caring for the patients about their perceptions of the usefulness, effectiveness, and relevance of the intervention. “Before we can move forward with a larger, randomized trial, we need to know the proportion of patients who are willing to enroll and complete the supportive oncology care at home intervention, how well this model of care will be received, and whether this shows promise for helping patients, their family caregivers, and their oncology team,” Nipp says.
He is, however, very hopeful. “With this pilot study, we hope to demonstrate feasibility and acceptability, which will help inform future trials to determine the efficacy of this supportive oncology care at home intervention,” Nipp adds. “We have learned a lot thus far, and we are excited for the potential we have seen to date.”
The study plans to recruit 20 patients overall, all of whom must be receiving care at MGH. Nipp believes that starting a trial like this with a relatively small group of patients with pancreatic cancer receiving neoadjuvant treatment prior to surgery makes the most sense for a pilot study.
“With this single arm pilot study, we will assess the feasibility and acceptability of a novel supportive oncology care at home intervention, which will inform future studies of this model of care, and we will use study results to develop a randomized trial in the future,” he says. “We are making progress in pancreatic cancer and neoadjuvant FOLFIRINOX represents a promising new paradigm. Thus, our efforts with this supportive oncology care at home intervention aim to improve the patient experience and enhance the quality of life and care for patients with pancreatic cancer receiving this novel paradigm of neoadjuvant treatment.”